For a better life
Wirbelsäulenimplantate direkt vom Hersteller
Prospitalia Kongress 2024 | Wir sehen uns in Frankfurt!
Der Prospitalia Kongress, mit dem diesjährigen Motto UMSICHT. WEITSICHT. AUSSICHT. GEMEINSAM ZUKUNFT SCHAFFEN, hat sich in den letzten Jahren als eine führende Plattform des Austauschs und Wissenstransfers im deutschen Gesundheitswesen etabliert. Hier bieten sich Möglichkeiten, sich über aktuelle Themen und Trends zu informieren, zu diskutieren und Erfahrungen auszutauschen.
Wir freuen uns, Sie dort zu treffen!
15. – 16. April 2024 im Kap Europa in Frankfurt

Frohe Ostern
Frohe Ostern von unserem Team an Sie und Ihre Familie! Mögen diese Feiertage voller Glück und wertvollen Momenten sein. Wir schätzen Ihre Treue und freuen uns auf eine weiterhin erfolgreiche Zusammenarbeit!

CadLab in der Anatomie der Universität Tübingen
Am 14. März 2024 fand unser CadLab in der Anatomie der Universität Tübingen statt. Wir freuen uns über den großen Erfolg dieser Veranstaltung! Themen des Kurses waren die operative Behandlung von thorakolumbalen Wirbelkörperfrakturen sowie die spinovelpine Stabilisierung am Humanpräparat. Mit erstklassigen Referenten konnten wir einen perfekten Mix aus Theorie und Praxis bieten.Herzlich bedanken möchten wir uns bei allen Teilnehmenden und bei unseren hervorragenden Referenten, Prof. Dr. med. Michael Rauschmann, Univ.-Prof. Dr. med. Erol Gercek und Dr. med. Erik Wegner für ihre Mitwirkung und die ausgewogenen Beiträge. Sie alle haben dazu beigetragen, diesen Tag zu einer herausragenden Plattform für den fachlichen Austausch von Wissen und Erfahrungen in der Wirbelsäulenchirurgie zu machen.
Das durchweg positive Feedback bestätigt uns darin, dass Fortbildung und der fachliche Austausch mit Experten von großer Bedeutung sind. Wir freuen uns auf weitere erfolgreiche und konstruktive Veranstaltungen - seien Sie gespannt!
Willkommen zum HumanTech CadLab-Seminar in der historischen Stadt Tübingen!
Freuen Sie sich auf eine inspirierende Fortbildung mit erstklassigen Referenten. Erleben Sie praxisnahe Einblicke in minimalinvasive Frakturversorgung und spinopelvine Stabilisierung der Wirbelsäule am Humanpräparat. Ein intensives Programm erwartet Sie - hochkarätige Vorträge, Hands-On-Training, spannende Diskussionen und wertvolles Networking. Dieses CadLab bietet nicht nur Wissenserweiterung, sondern auch eine einzigartige Möglichkeit zum Erfahrungsaustausch mit Kollegen und Experten. Wir freuen uns darauf!Events, Kongresse und Seminarreihe 2024 – SAVE THE DATE
HumanTech Spine GmbH plant im Jahr 2024 nicht nur eine spannende Seminarreihe, sondern auch diverse nationale und internationale Messeauftritte. Unsere Veranstaltungsreihe bietet einen einzigartigen Einblick in die neuesten Entwicklungen der Medizintechnikbranche, während unsere Messeauftritte eine ideale Gelegenheit bieten, unser Team zu treffen und unsere wegweisenden Produkte und Technologien hautnah zu erleben. Wir laden Sie herzlich dazu ein!
Upcoming Events

Ein unvergessliches Messeerlebnis in unserer Heimatstadt!
Der diesjährige DWG Kongress in Stuttgart war für uns ein absolutes Highlight. Wir möchten uns herzlich bei Ihnen allen bedanken! Die Vielzahl an interessanten Gesprächen und positiven Begegnungen haben deutlich gemacht, wie lebendig und vielseitig unsere Community ist. Ihr aufrichtiges Interesse an unserem Unternehmen hat uns nicht nur motiviert, sondern auch inspiriert. Wir schauen bereits voller Vorfreude auf kommende Events.
Ein Rundum-Erfolg!
Wir möchten uns von ganzem Herzen bei den zahlreichen nationalen und internationalen Teilnehmern unserer Fortbildungsveranstaltung der HuT Akademie am Institut für Anatomie in Erlangen bedanken. Wieder einmal war es ein lebendiges, dynamisches und erfolgreiches Event. Es war inspirierend zu sehen, wie wir in einer positiven Lernumgebung gemeinsam Wissen geteilt und neue Perspektiven entdeckt haben. Danke an alle Mitwirkenden! Ein besonderer Dank geht an unsere hochgeschätzten Referenten, die durch ihre Fachkompetenz und ihre lebendige Präsentation zu einer erstklassigen Wissensvermittlung beigetragen haben.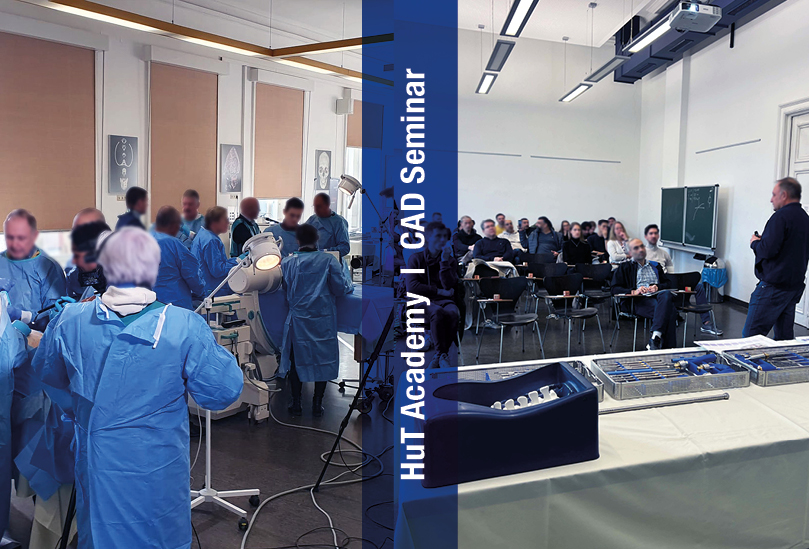
EUROSPINE 2023 | Frankfurt | Germany
Ein erfolgreicher Messeauftritt liegt hinter uns!Wir sind wieder einmal überwältigt von den zahlreichen Besuchern, den vielen wertvollen Gesprächen und den schönen Begegnungen auf unserem Messestand bei der diesjährigen Eurospine in Frankfurt. Ihre Fragen und Anregungen sowie der Wissensaustausch sind für uns von unschätzbarem Wert – danke für Ihre Zeit, Ihr Interesse und Ihr Vertrauen in uns. Wir freuen uns auf weitere zukünftige Begegnungen!

Erfolgreich hat die Fortbildungsveranstaltung der Akademie der DWG am Institut der Anatomie II in Frankfurt a.M. stattgefunden. An den Präparat-Workshops präsentierte HumanTech das Wirbelkörperersatzsystem Samson und das minimalinvasive Fixationssystem Venus Mini 2.0. Wir bedanken uns bei den zahlreichen aktiven Teilnehmer und bei allen Mitwirkenden für den großartigen Erfahrungsaustausch und die professionelle Durchführung der Basiskurse.


Premiere in Essen
Unter der Organisation von ProSympos GmbH hat kürzlich in Essen erfolgreich eine Fortbildung unserer HUT Akademie stattgefunden. Mit großem Interesse und viel Teamspirit wurden die Teilnehmer von Dr. med. Daniel Seeger und Dr. med. Ercan Sagnak umfassend zu den Themen „Technik der lumbopelvine Stabilisierung bei Sakrumfrakturen und minimalinvasive Verlängerung nach kranial“ geschult. Wir möchten uns bei allen Mitwirkenden für die professionelle Durchführung, den großartigen Erfahrungsaustausch und die konstruktiven Beiträge bedanken.Essen – wir kommen wieder!
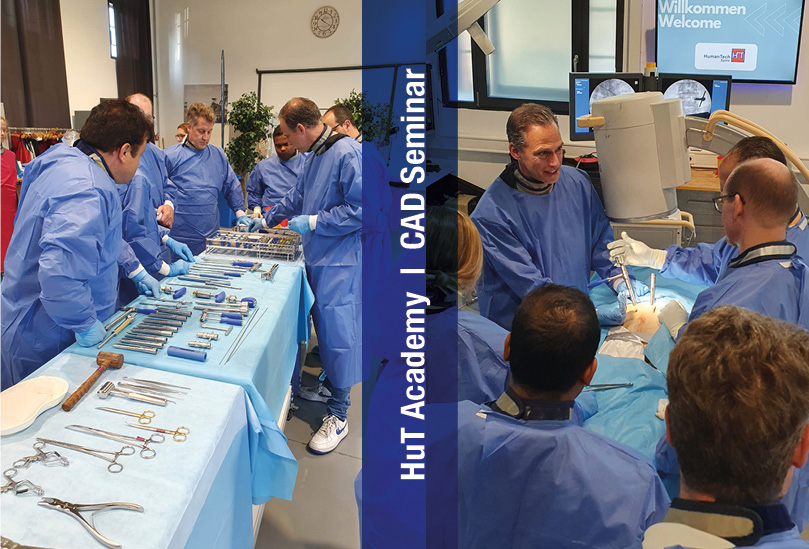
Wir sind begeistert!
Die erste zertifizierte Fortbildung unserer HUT Akademie hat kürzlich in Tübingen stattgefunden. Die zahlreichen nationale und internationalen Teilnehmer wurden von erfahrenen Anwendern rund um das Thema „Wirbelkörperersatz inkl. perkutane Instrumentierung“ intensiv geschult. Begeisterung, Teamspirit, Weiterbildung - danke für den großartigen Erfahrungsaustausch, die interessanten Begegnungen und die konstruktiven Beiträge.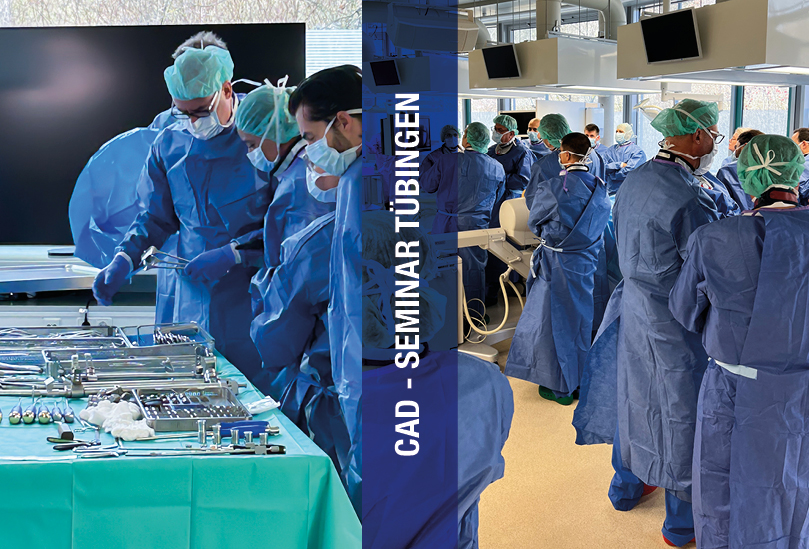
CadLab-Seminare

Prof. Rauschmann (SANA-Klinik Offenbach) und Dr. Krebs (OKM Markgröningen) wussten die zahlreichen Teilnehmer der Live-Übertragung aus der Universität Erlangen zu begeistern. Mit Dr. Hassel (Loretto-Klinik Freiburg) und Dr. Sagnak (DKH Hamburg) nahmen zusätzlich 2 außerordentlich erfahrene Referenten mit ihren Online-Vorträgen teil.
HumanTech freut sich, dass diese Art von Live-Vorträgen von Cad-Labs immer beliebter wird und sich von den zunehmenden, manchmal wirklich langweiligen "Marketing Webinars" abhebt. Die von HumanTech unterstützten Spinetraining-Veranstaltungen erhalten nun auch von den deutschen Ärztekammern eine hervorragende Anerkennung.
 CadLab - Seminare zu verschiedenen Themen, wie z.B. „Wirbelkörperersatz oder minimalinvasive Fixation“
CadLab - Seminare zu verschiedenen Themen, wie z.B. „Wirbelkörperersatz oder minimalinvasive Fixation“ Vertriebspartner gesucht!











